
- [email protected]
+91 40 2344 5962 - View Stock Quote

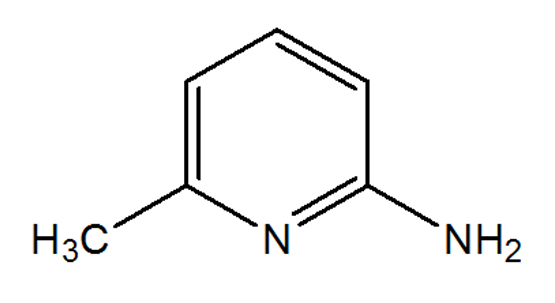

Synonyms : 2-Amino-6-Picoline; 6-Methyl-2-Pyridinamine; 6-Amino-2-Picoline
CAS No. : [1824-81-3]
Molecular Formula : C6H8N2
Formula weight : 108.14
2-Amino-6-methylpyridine is used as an intermediate in the pharmaceutical industry for the manufacture of nalidixic acid etc. It is also used for laboratory utilization, analysis, research and fine chemistry.
2-Amino-6-Methylpyridine is another organic compound which is basically considered as alkaline. It has very faint turbidity in water.
This compound is basically obtained by reacting ?-picoline with ammonia in the presence of cobalt-containing catalysts. Nevertheless, only very moderate yields of 2-amino-6-methylpyridine can be obtained in this reaction.
2-amino-6-methylpyridine can also be prepared from ?-picoline and chloramine. The only disadvantages of this reaction are the use of chloramine, which is inclined towards decomposing and is therefore quite difficult to handle. In addition to this, the isolation of the ?-amino-?'-picoline in multi-stage is a cumbersome process.
When the ?-amino-pyridines is prepared through the reaction of the corresponding hydroxy compounds with ammonia is actually known from German Offenlegungsschrift. The catch in the above two processes are that in the first case of 2-amino-6-methylpyridine, the amination step proceeds with only a 27% yield. In the second case, the difficulty is the availability and ready accessibility of the ?-alkyl-?'-hydroxypyridines which are used as the starting substances for the reaction.
Another important fact regarding this is that the reaction of ammonia and phenol over ZSM-5 zeolites at 510° C to give, besides aniline and a minor amount of ?-picoline. Besides, a relatively large number of by-products occurs in this reaction like toluidines, xylidines and diphenylamine. The formation of these by-products can be evidently attributed to a very high-temperature reaction. Thus, it is generally associated with high industrial expenditure which ultimately leads to coking and frequent change or regeneration of the catalyst.
2-Amino-6-methylpyridine finds a wide application as an intermediate in the pharmaceutical industry for the manufacture of nalidixic acid etc. It is also evidently used for laboratory utilization, analysis, research and fine chemistry.